Ozurdex drug delivery implant for eyes

Ozurdex drug delivery implant for eyes
Ozurdex is the first FDA approved, bio-degradable drug delivery system for the eye.
Ozurdex is a drug-filled implant injected into the eye’s vitreous, the gel-like substance filling the inside of the eyeball.
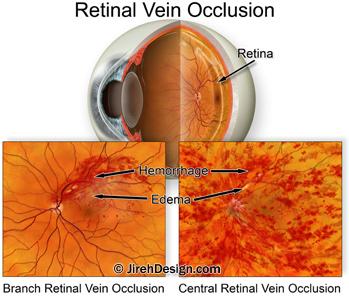
The (corticosteroid) drug-filled implant is approved for treating adults with macular edema (swelling) caused by retinal vein occlusion and diabetic macular edema, caused by diabetic retinopathy. It is also used to treat the inflammatory effects of uveitis.
What is Ozurdex?
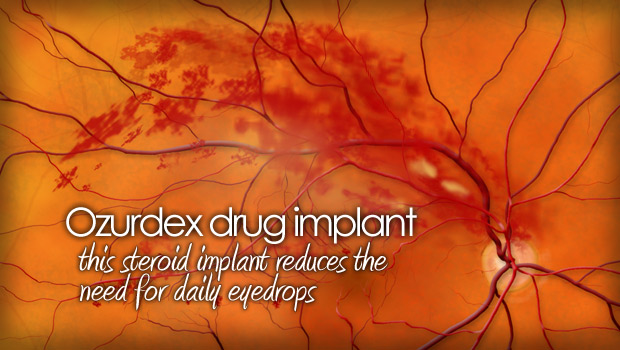
An intraocular steroid known to reduce swelling in the macula. This reduction in swelling may help reverse vision loss. The implant doesn’t ever need to be removed because it slowly dissolves over time inside the eye, releasing its medication.
How does Ozurdex work?
The tiny rod-shaped implant is injected into the eye through a very small needle-like application device. While in the eye, the implant dissolves into the vitreous for up to 6 months, reducing the need for multiple eye drops every day.
Are their any risks or restrictions with Ozurdex?
Due to the long-term nature of the implanted medication, there is a strict protocol for its use. The implant should not be performed on a patient who has any infections of the eye or surrounding eye area. This medication should also not be used in patients with advanced glaucoma or patients with a known sensitivity to intraocular steroids.
Ozurdex side effects
The most common side effects reported in patients include: increased eye pressure, minor bleeding in the “whites” of the eye, eye pain, cataract, vitreous detachment and headache. These side effects should be monitored closely.
Dr. Deupree has been using Ozurdex on select cases since 2009. His patients have shown improved symptoms and reduced retinal vein occlusion and diabetic retinopathy related edema.
Mentioned in this article: Pred Forte, eye drops, diabetic retinopathy.