Lucentis treatment for eye disease
Lucentis eye injection for macular degeneration and other eye diseases Lucentis is used to treat age-related macular degeneration (AMD) and diabetic […]


Lucentis treatment for eye disease
Lucentis eye injection for macular degeneration and other eye diseases Lucentis is used to treat age-related macular degeneration (AMD) and diabetic […]
Lucentis eye injection for macular degeneration and other eye diseases
Lucentis is used to treat age-related macular degeneration (AMD) and diabetic macular edema – leading causes of vision loss.
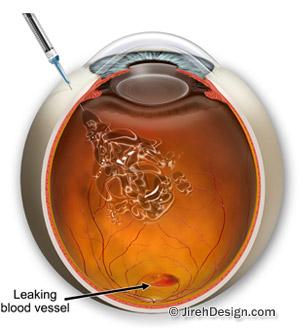
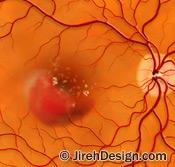
Lucentis (generic: ranibizumab) treats the wet form of macular degeneration. There are two types of macular degeneration: dry and wet. In the “wet” form of AMD, abnormal blood vessels grow under the retina, in the back of the eye. Sometimes these vessels leak blood or fluid that causes blurred or distorted vision. Without treatment, vision loss may be quick and severe. This is called “wet” macular degeneration, as opposed to dry AMD.
Lucentis eye injection for macular degeneration
This medication works by inhibiting the growth of the abnormal blood vessels that cause AMD. It is also used to treat swelling of the macula due to diabetic macular edema. The goal of treatment is to prevent further loss of vision. Although some patients have regained vision, the medication may not restore vision that has already been lost, and may not ultimately prevent further loss of vision caused by the disease.
How is Lucentis eye injection used?
After the pupil is dilated and the eye is numbed with special drops and injectables, the medication is injected into the vitreous, or jelly-like substance in the inside chamber of the eye. The medicine is administered by an injection into your eye as needed at regular intervals (about every four to six weeks) – your eye doctor will tell you how often you will need to receive the injection.
Lucentis alternatives
You do not have to receive treatment for your condition, although without treatment, AMD can lead to further vision loss and legal blindness, sometimes very quickly. Other forms of treatment are available. There are other FDA-approved treatments for wet AMD: photodynamic therapy with a drug called Visudyne and injection into the eye with other drugs called anti-VEGFs: Avastin, Eylea and Macugen. Although all of these treatments have been proven to slow down the rate of visual loss, patients may not get back better vision.
In addition to the FDA-approved medications, some eye doctors use other medications that were not specifically approved for use in the treatment of AMD, but which have shown some benefit. One of the medications used this way is intravitreal Kenalog.
Lucentis side effects and complications
New Lucentis vs. Stroke Information
A study showed that Ranibizumab use in stroke patients may lead to an increased risk of stroke.
However, once the final study was complete, results showed that there was no associated risk of stroke in patients getting Ranibizumab treatment.
Whenever a medication is used in a large number of patients, a small number of coincidental life-threatening problems may occur that have no relationship to the treatment. For example, patients with diabetes are already at increased risk for heart attacks and strokes. If one of these patients being treated with Lucentis suffers a heart attack or stroke, it may be caused by the diabetes and not the Lucentis treatment.
Watch the “How vision works” animation
Risks of intravitreal eye infections
Your condition may not get better or may become worse. Any or all of the following complications may cause decreased vision and/or have a possibility of causing blindness. Additional procedures may be needed to treat these complications. During the follow up visits or phone calls, you will be checked for possible side effects and the results will be discussed with you.
Possible complications of the procedure and use of Ranibizumab include but are not limited to: retinal detachment, cataract formation (clouding of the lens of the eye), glaucoma (increased pressure in the eye), hypotony (reduced pressure in the eye), damage to the retina or cornea, and bleeding.
There is also the possibility of an eye infection (endophthalmitis). Your eye doctor should take specific precautions to reduce the possibility of this occurring. Any of these rare complications may lead to severe, permanent loss of vision. Side effects may include eye pain, subconjunctival hemorrhage (bloodshot eye), vitreous floaters, irregularity or swelling of the cornea, inflammation of the eye, and visual disturbances such as small specks in the vision.
Click here to watch a Lucentis treatment animation
Most of this page’s information provided by: Ophthalmic Mutual Insurance Company
See also:
Click here to watch a Lucentis treatment animation
Avastin eye injection
Eylea eye injection
Macugen eye injeciton
Macular degeneration
Diabetic retinopathy
Central retinal vein occlusion