Avastin treatment for eye disease
Avastin treatment is an in-office procedure for macular degeneration, diabetic retinopathy and retinal vein occlusion. Here we discuss how Avastin (Bevacizumab) eye injection is used and why.


Avastin treatment for eye disease
Avastin treatment is an in-office procedure for macular degeneration, diabetic retinopathy and retinal vein occlusion. Here we discuss how Avastin (Bevacizumab) eye injection is used and why.
What is Avastin?
Eye doctors use Avastin to treat several eye and retina diseases, like age-related macular degeneration (AMD). AMD is the leading cause of blindness in people over 50 years of age in the United States. AMD is caused by the breakdown of the central portion of the retina, called the macula (the highly sensitive part of your eye that works like the film in a camera).
Avastin also treats diabetic macular edema and central retinal vein occlusion (CRVO).
The macula is responsible for the detailed central vision in the eye. The macula enables us to see fine print, recognize faces and drive our car. There are two types of macular degeneration: dry and wet.
In the wet form of macular degeneration, abnormal blood vessels grow in the back of the eye. Some of these vessels leak blood or fluid. This leakage causes blurred or distorted vision. Without treatment, vision loss can be quick and severe.
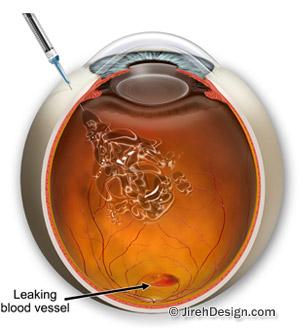
Chronic macular edema, or swelling around the macula, is a condition that affects vision but does not respond well to the usual treatment drugs, like eye steroids. It can occur with conditions such as central retinal vein occlusion and diabetic retinopathy. Without effective treatment, vision loss could get worse or become permanent.
What is Avastin off-label use?
Avastin was not initially developed to treat eye conditions. Based upon the results of clinical trials demonstrating its safety and effectiveness, Avastin was approved as a chemotherapy drug by the U.S. Food and Drug Administration (FDA) for the treatment of metastatic colorectal cancer. As a condition of approval, the manufacturer produced a “label” explaining the indications, risks, and benefits. The label explains that Avastin works by blocking a substance known as vascular endothelial growth factor or VEGF. Blocking or slowing VEGF helps prevent further growth of the blood vessels that the cancer needs to continue growing.
Once the FDA approves a device or medication, physicians may use it “off-label” for other purposes. Ophthalmologists are using Avastin “off-label” to treat AMD and similar conditions because research indicates that VEGF is one of the causes for the growth of the abnormal vessels that cause these conditions. Most patients who receive these treatments have less fluid and more normal-appearing maculas, and enjoy improved vision. Bevacizumab also treats macular edema in some diabetic patients and cases of retinal vein occlusion.
Watch the “How vision works” animation
Off-label drugs
Here are some examples of common drugs being used “off-label”:
1) Eye antibiotics
2) Botox
3) Zoloft & Paxil
4) Methotrexate
Possible Avastin limitations
The goal of Avastin treatment is to prevent further loss of vision. Although some patients regain vision, the medication may not restore vision that was already lost. Avastin may not ultimately prevent further loss of vision from the eye disease.
 How is Avastin eye injection done?
How is Avastin eye injection done?
After the doctor dilates the pupil and numbs the eye with anesthesia, he injects the medication into the vitreous — the jelly-like substance in the back chamber of the eye. To demonstrate this, you can watch an Avastin injection animation here. Your doctor may continue to use Avastin injection into your eye as needed at regular intervals (about every four to six weeks). Your ophthalmologist will tell you how often you will need to receive the injection, and for how long.
Avastin alternatives
You do not have to receive treatment for your condition but without treatment, these diseases can lead to further vision loss and blindness, sometimes very quickly. Other forms of treatment are available. At present, there are several other FDA-approved treatments for wet age-related macular degeneration: photodynamic therapy with a drug called Visudyne and injection into the eye of other VEGF inhibitors drugs called Eylea, Macugen and Lucentis.
Although all of these treatments are proven to slow down the rate of visual loss, many people do not get back better vision. Dr. Deupree will discuss with you the benefits and risks of these optional treatments.
In addition to the FDA-approved medications, some ophthalmologists use intravitreal Kenalog, a long-acting anti-inflammatory drug, to treat eye conditions like yours.
Avastin side effects and complications
Complications when Avastin is given to patients with cancer
When Avastin is given to patients with colorectal cancer, a small amount of patients experience serious and sometimes life-threatening complications.
Patients who experienced these complications not only had colon cancer, but were also given 400 times the dose typically given in the eye. Also, cancer patients receive treatments at more frequent intervals, and in a way (intravenous) that spread the drug throughout their bodies.
Avastin eye injection risks
Ophthalmologists believe that the risk of these complications for patients with eye conditions is low. On average, patients who receive Avastin for eye conditions are typically healthier than the cancer patients, and receive a significantly smaller dose only inside their eye. There are ongoing clinical trials of similar drugs as well as studies of patients getting Avastin “off-label.” One study of patients treated with Avastin intravenously reported only a mild elevation in blood pressure. Another study of patients treated with intravitreal Avastin (Bevacizumab injected into the eye) did not have these elevations or the other serious problems seen in the patients with cancer.
It is important to understand that the benefits and risks of intravitreal Avastin for eye conditions are not yet fully known. In addition, whenever a medication is used in a large number of patients, a small number of coincidental life-threatening problems may occur that have no relationship to the treatment.
For example, patients with diabetes are already at increased risk for heart attacks and strokes. If one of these Avastin patients suffers a heart attack or stroke, the cause could be the diabetes and not the Avastin.
Side effects of intravitreal eye injections
Your condition may not get better or may even become worse. Any or all of these complications may cause a decrease in vision and/or legal blindness. Treating these complications could require additional procedures. During the follow up visits or phone calls, your doctor or eye technician will discuss possible side effects.
Any medication has the potential to cause allergic reactions in a small number of people. Symptoms of an allergic reaction can include a rash, hives, itching, shortness of breath, and rarely, death (extremely rare). In general, allergic reactions to medicines are more likely to occur in people who have allergies to other drugs, foods, or things in the environment, such as dust or grass. If you have allergies to other medicines, foods, or other things in the environment, or if you have asthma, you should let Dr. Deupree or his staff know.
Possible complications and side effects of the procedure and administration of Avastin include but are not limited to retinal detachment, cataract formation (clouding of the lens of the eye), glaucoma (increased pressure in the eye), hypotony (reduced pressure in the eye), damage to the retina or cornea (structures of the eye), and bleeding.
There is also the possibility of an eye infection (endophthalmitis). Any of these rare complications may lead to severe, permanent loss of vision.
Patients who receive an injection of Avastin may experience less severe side effects related to the preparation procedure. These side effects may include eye pain, subconjunctival hemorrhage (bloodshot eye), vitreous floaters, irregularity or swelling of the cornea, inflammation of the eye, and visual disturbances.
Click here to watch an Avastin injection animation
Illustrations and animations by www.JirehDesign.com
See also…
Click here to watch a Lucentis treatment animation
Lucentis eye injection
Eylea eye injection
Macugen eye injeciton
Macular degeneration
Diabetic retinopathy
Central retinal vein occlusion
Some of this page's information provided by: Ophthalmic Mutual Insurance Company